Chemical BondingHard
Question
In a closed packed structure of mixed oxides, the lattice is composed of mixed oxides ions. One-eighth of tetrahedral voids are occupied by divalent cations (A2+) while one-half of octahedral voids are occupied by trivalent cations (B3+). The formula of mixed oxide is
Options
A.A2BO3
B.AB2O4
C. A2BO4
D.AB2O4
Solution
For close-packed structure (fcc type)
Number of O2- = 4/unit cell
Number of A2+ =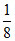 × TV =
× TV = 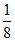 × 8 = 1
× 8 = 1
Number of B3+ =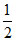 × OV =
× OV = 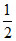 × 4 = 2
× 4 = 2
Formula: A12+ B23+ O42- ⇒ AB2O4
Number of O2- = 4/unit cell
Number of A2+ =
Number of B3+ =
Formula: A12+ B23+ O42- ⇒ AB2O4
Create a free account to view solution
View Solution FreeMore Chemical Bonding Questions
Given below are two statements :Statement I: The halogen that makes longest bond with hydrogen in HX, has the smallest c...The correct order of increasing X-O-X bond angle is (X = H, F or Cl) :-...Given below are two statements:Statement I: Among $\left\lbrack Cu\left( {NH}_{3} \right)^{2 +},\left\lbrack Ni(en)_{3} ...The bond angle between H-O-H in ice is closest to...In which of the following compounds, breaking of covalent bond take place :-...