ThermochemistryHard
Question
Ethanol can undergoes decomposition to form two sets of products.
C2H5OH (g) →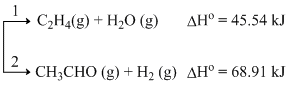
If the molar ratio of C2H4 to CH3CHO is 8 : 1 in a set of product gases, then the energy involved in the decomposition of 1 mole of ethanol is
C2H5OH (g) →
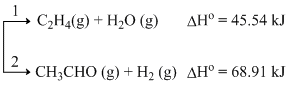
If the molar ratio of C2H4 to CH3CHO is 8 : 1 in a set of product gases, then the energy involved in the decomposition of 1 mole of ethanol is
Options
A.65.98kJ
B.48.137 kJ
C.48.46 kJ
D.57.22 kJ
Solution
ᐃr H = 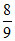 × 45.54 +
× 45.54 + 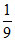 × 68.91 = 48.137 kJ.
× 68.91 = 48.137 kJ.
Create a free account to view solution
View Solution FreeMore Thermochemistry Questions
The thermochemical equation for the dissociation of hydrogen gas into atoms may be written asH2 → 2H; ΔH = 432.0 kJ.What...Among the following the reaction for which ΔH = ΔE is...For the given reactionsSiO2 + 4HF → SiF4 + 2H2O, ᐃH = -10.17 kcalSiO2 + 4HCl → SiCl4 + 2H2O, ᐃH ...A volume of 4.0 L of a mixture of ethane and methane gases on complete combustion at 300 K produced 6.0 L of carbon diox...In a flask, colourless N2O4 is in equilibrium with brown coloured NO2. At equilibrium, when the flask is heated at 373 K...