Chemical BondingHard
Question
Which of the following species contain three bond pairs and one lone pair around the central atom ?
Options
A.H2O
B.BF3
C.NH2-
D.PCl3
Solution
(a) H2O ⇒ 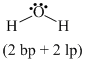
(b) BF3 ⇒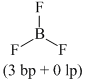
(c) NH2- ⇒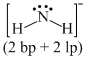
(d) PCl3 ⇒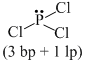
Thus, in PCl3, the central atom P has three bond pairs and one lone pair.
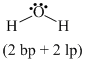
(b) BF3 ⇒
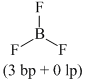
(c) NH2- ⇒
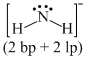
(d) PCl3 ⇒
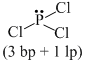
Thus, in PCl3, the central atom P has three bond pairs and one lone pair.
Create a free account to view solution
View Solution FreeMore Chemical Bonding Questions
The species in which the central atom uses sp2 - hybrid orbitals in its bonding is :...The helical structure of proteins is stabilised by:-...Which of the following combination(s) lead(s) to the formation of gerade molecular orbital(s) -...Which one of the following compounds is a peroxide ?...Match the lists I and II and write the correct matching :-...