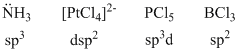JEE AdvancedChemical BondingHard
Question
The correct order of hybridisation of the central atom in the following species NH3, [PtCl4]2-, PCl5 and BCl3 is :
Options
A.dsp2, dsp3, sp2 amd sp3
B.sp3, dsp2, sp3d amd sp2
C.dsp2, sp2, sp3 dsp3
D.dsp2, sp3, sp2 dsp3
More Chemical Bonding Questions
....................(1) ....................(2)If both the reaction shows kinetic isotope effect then...Which of the following statements is true ?...The dipole moment of AX3, BX3 and CY3 are 4.97 × 10-30, 0.60 × 10-30 and 0.00 C.M. respectively, then the shap...The correct order of increasing C - O bond lenght of CO, CO32-, CO2 is :...Correct order of basic strength of this compound is...