Chemical BondingHard
Question
The correct order of increasing C - O bond lenght of CO, CO32-, CO2 is :
Options
A.CO32- < CO2 < CO
B.CO2 < CO32- < CO
C.CO < CO32- < CO2
D.CO < CO2 < CO32-
Solution
Bond lenght 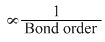
On the basis of M. O theory
Bond order of CO, CO2 and CO32- (considering resonance) are 3, 2 and 1.33 respectively thus order of bond length is CO < CO2 < CO32-
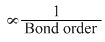
On the basis of M. O theory
Bond order of CO, CO2 and CO32- (considering resonance) are 3, 2 and 1.33 respectively thus order of bond length is CO < CO2 < CO32-
Create a free account to view solution
View Solution Free