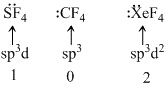Chemical BondingHard
Question
Molecular shape of SF4, CF4 and XeF4 are :
Options
A.Thr same, with 2, 0 and 1 lone pair of electrons respectively
B.Thr same, with 1, 1, 1, lone pair of electrons respectively
C.different, with 0, 1, and 2 lone pair of electrons respectively
D.different, with 1, 0, and 2 lone pair of electrons respectively
More Chemical Bonding Questions
The intermolecular interaction that is dependent on the inverse cube of distance between the molecules is :-...The heat of atomization of methane and ethane are 360 kJ/mol and 620 kJ/mol, respectively. The longest wavelength of lig...The helical structure of proteins is stabilised by:-...The structure of IF7 is :...Match the following graphs :-GraphA (i) (+ deviation)B (ii) IdealC (iii) (-) deviation...