JEE Main | 2014Chemical BondingHard
Question
For which of the following molecule significant μ ≠ 0?
(a)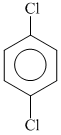 (b)
(b) 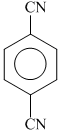
(c)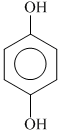 (d)
(d) 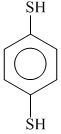
(a)
 (b)
(b) 
(c)
 (d)
(d) 
Options
A.Only (a)
B.(a) and (b)
C.Only (c)
D.(c) and (d)
 (b)
(b) 
 (d)
(d) 