AIPMT | 2015Chemical BondingHard
Question
Which of the following species contains equal number of σ - and π- bonds :-
Options
A.XeO4
B.(CN)2
C.CH2(CN)2
D.HCO3
Solution
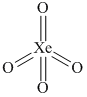
number of σ bonds = 4
number of π bonds = 4
Create a free account to view solution
View Solution FreeMore Chemical Bonding Questions
For binary hydride of formula MXn. The value of n can be fractional per :-...Which one of the following pairs of molecules will have permanent dipole moments for both members?...A magnetic moment of 1.73 BM will be shown by one among the following :-...Which of the following solid has maximum melting point :-...Which of the following is correct graph for the reaction ?...