ThermodynamicsHard
Question
Calculate ᐃfGo for (NH4CI, s) at 310 K.
Given : ᐃfHo (NH4CI, s) = - 314.5 kJ/mol; ᐃrCp = 0
SoN2(g) = 192 JK-1 mol-1; SoH2(g) = 130.5 JK-1 mol-1;
All given data are at 300 K.
Given : ᐃfHo (NH4CI, s) = - 314.5 kJ/mol; ᐃrCp = 0
SoN2(g) = 192 JK-1 mol-1; SoH2(g) = 130.5 JK-1 mol-1;
All given data are at 300 K.
Options
A.-198.56 kJ/mol
B.-426.7 kJ/mol
C.-202.3 kJ/mol
D.None of these
Solution
ᐃfSo(NH4CI,s) at 300 K (300 K ᐃfSo(NH4CI,s))
= SoNH4Cl(s) -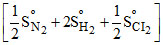
= - 374 JK-1 mol-1
ᐃfCP = 0
∴ ᐃfSo310 = ᐃfSo300
= - 374 JK-1 mol-1
ᐃrHo310 = ᐃrHo300 = - 314.5
ᐃfGo310 = ᐃfHo - 310ᐃSo = - 314.5 -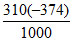 = - 198.56 kJ/mol.
= - 198.56 kJ/mol.
= SoNH4Cl(s) -
= - 374 JK-1 mol-1
ᐃfCP = 0
∴ ᐃfSo310 = ᐃfSo300
= - 374 JK-1 mol-1
ᐃrHo310 = ᐃrHo300 = - 314.5
ᐃfGo310 = ᐃfHo - 310ᐃSo = - 314.5 -
Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
Standard entropy of X2, Y2 and XY2 are 60, 40 and 50 JK-1mol-1, respectively. For the reaction,, ᐃH = - 30 kJ, to ...For a spontaneous process, entropy:...In which of the following reactions, standard reaction entropy change (ᐃSo) is positive and standardGibb′s e...In which of the following reactions, standard reaction entropy changes (ᐃSo) is positive and standard Gibb′s...ᐃSfusion of a substance is ′x′ and ᐃHvap is ′y′, then ᐃHsublimation will be...