NEETThermochemistryHard
Question
The enthalpy of combustion of propane (C3H8) gas in terms of given data is :
Bond energy (kJ/mol)
εC-H εO=O εC=O εO-H εC-C
+x1 +x2 +x3 +x4 +x5
Resonance energy of CO2 is - zkJ/mol and ᐃHvaporization [H2O(l)] is ykJ/mol.
Bond energy (kJ/mol)
εC-H εO=O εC=O εO-H εC-C
+x1 +x2 +x3 +x4 +x5
Resonance energy of CO2 is - zkJ/mol and ᐃHvaporization [H2O(l)] is ykJ/mol.
Options
A.8x1 + 2x5 + 5x2 - 6x3 - 8x4 - 4y - 3z
B.6x1 + x5 + 5x2 - 3x3 - 4x4 - 4y - 3z
C.8x1 + 2x5 + 5x2 - 6x3 - 8x4 - y - z
D.8x1 + x5 + 5x2 - 6x3 - 8x4 - 4y + 3z
Solution
C3H8(g) + 5O2 → 3CO2(g) + 4H2O(l)
ᐃCH =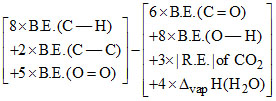
ᐃCH =
Create a free account to view solution
View Solution FreeMore Thermochemistry Questions
The enthalpy change (ᐃH) the reaction, N2(g) + 3H3(g)   → 2NH3(g) is - 92.38 kJ at 398 K. The internal e...Calculate the standard free energy change for the ionization HF(aq) → H+ (aq) + F– (aq) from the following data.HF(aq) →...The enthalpy of formation of KCl(s) from the following data is(i) KOH(aq) + HCl(aq) → KCl(aq) + H2O(l): ΔH = −13.7 kcal(...Which of the following molecules will have different values of standard molar enthalpy of formation, one calculated usin...At 300 K, the standard enthalpies of formation of C6H5COOH(s), CO2(g) and H2O(l) are −408, −393 and −286 kJ/mol, respect...