ThermochemistryHard
Question
Oxidising power of chlorine in aqueous solution can be determined by the parameters indicated below:
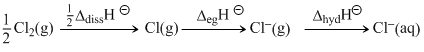
The energy involved in the conversion of Cl2(g) to Cl-(g)
Cl2(g) to Cl-(g)
(using the data, ᐃdissHCl2- = 240 k J mol-1, ᐃegHCl- = - 349 k J mol-1 ᐃhydHCl- = - 381 k J mol -1) will be
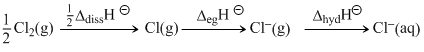
The energy involved in the conversion of
 Cl2(g) to Cl-(g)
Cl2(g) to Cl-(g)(using the data, ᐃdissHCl2- = 240 k J mol-1, ᐃegHCl- = - 349 k J mol-1 ᐃhydHCl- = - 381 k J mol -1) will be
Options
A.+152 kJmol-1
B.-610 kJmol-1
C.-850 kJmol-1
D.+ 120 kJmol-1
Solution
For the process 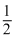 Cl2(g) → Claq-
Cl2(g) → Claq-
ᐃH =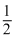 ᐃHdiss of Cl2 + ᐃegCl + ᐃhydCl-
ᐃHdiss of Cl2 + ᐃegCl + ᐃhydCl-
= +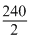 - 349 - 381
- 349 - 381
= - 610 kJ mol-1
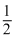 Cl2(g) → Claq-
Cl2(g) → Claq-ᐃH =
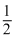 ᐃHdiss of Cl2 + ᐃegCl + ᐃhydCl-
ᐃHdiss of Cl2 + ᐃegCl + ᐃhydCl- = +
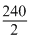 - 349 - 381
- 349 - 381= - 610 kJ mol-1
Create a free account to view solution
View Solution FreeMore Thermochemistry Questions
Reaction involving gold have been of particular intersect to alchemists. Consider the following reactions, Au(OH)3 + 4 H...Calculate proton affinity of NH3(g) from the following data.ΔHdissociation H2 = 218 kJ mole–1ΔHdissociation Cl2 = 124 kJ...Reactions involving gold have been of particular interest to a chemist. Consider the following reactions.Au(OH)3 + 4HCl ...From the following thermochemical equations, find out the bond dissociation enthalpy of CH3−H bond.CH3I(g) → CH3(g) + I(...The standard molar enthalpies of formation of trinitrotoluene(l), CO2(g) and H2O(l) are 65, −395 and −285 kJ/mol, respec...