Chemical BondingHard
Question
The species in which the central atom uses sp2 - hybrid orbitals in its bonding is :
Options
A.PH3
B.NH3
C.CH3+
D.SbH3
Solution
In CH3+, three C - H bond(sp2 - sσ bonds) are arranged in planar triangular manner and one unhybridised p-orbital remain 1 to the plane
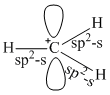
Create a free account to view solution
View Solution FreeMore Chemical Bonding Questions
For which of the following molecule significant μ ≠ 0? (a) (b) (c) (d)...Which of the following comound is covalent ?...If a molecule MX3 has a zero dipole moment, the sigma bonding orbitals used by M (atomic number < 18) :-...Cu+ + e- → Cu Eo = a1 voltCu+2 + 2e- → Cu Eo = a2 voltthen for Cu2+ + e- → Cu+, Eo will be :-...Dipole moment is shown by :...