Chemical BondingHard
Question
Of the following compounds, which will have a zero dipole moment ?
Options
A.1, 1-dichloroethylene
B.cis - 1, 2- dichloroethylene
C.trans 1, 2- dichloroethylene
D.None of these
More Chemical Bonding Questions
The correct statement(s) regarding, (i) HClO, (ii) HClO2, (iii) HClO3 and (iv) HClO4, is(are)...Ortho-Nitrophenol is less soluble in water than p- and m- Nitrophenols because :-...The pair of species with the same bond order is...Bond Angle is maximum in :-...The molecule that has linear structure is :...
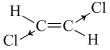 ; μR = 0
; μR = 0