Chemical BondingHard
Question
Ortho-Nitrophenol is less soluble in water than p- and m- Nitrophenols because :-
Options
A.Melting point of o-Nitrophenol is lower than those of m- and p- isomers
B.o-Nitrophenol is more volatile in steam than those of m- and p- isomers
C.o-Nitrophenol shows Intramolecular H-bonding
D.o-Nitrophenol shows Intermolecular H-bonding
Solution
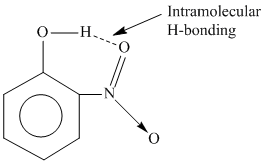
due to presence of intramolecular H-bonding its interaction with water molecule will be poor, resulting into less solubility of the compound. While para & meta derivative forms intermolecular H-bonding with water.
Create a free account to view solution
View Solution FreeMore Chemical Bonding Questions
Which is the strongest acid in the following ?...The shape of NH3 molecule is :...Amongst LiCl, RbCl BeCl2 and MgCl2, the compounds with the greatest and the least jonic character, respectively are :-...Which artificial sweetener contains chlorine ?...The following compounds have been arranged in order of their increasing thermal stability ties. Identify the current ord...