Chemical EquilibriumHard
Question
(i) N2(g) + O2(g) ⇋ 2NO(g), K1
(ii)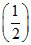 N2(g) +
N2(g) + 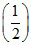 O2(g) ⇋ NO(g) ; K2
O2(g) ⇋ NO(g) ; K2
(iii) 2NO(g) N2(g) + O2(g) ; K3
(iv) NO(g) ⇋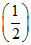 N2(g) +
N2(g) + 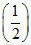 O2(g) ; K4
O2(g) ; K4
Correct relation between K1, K2 and K4 is/are :
(ii)
(iii) 2NO(g) N2(g) + O2(g) ; K3
(iv) NO(g) ⇋
Correct relation between K1, K2 and K4 is/are :
Options
A.K1 × K3 = 1
B.√K1 × K4 = 1
C.√K3 × K2 = 1
D.None
Solution
From given reactions
(i) = - (iii) ;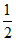 (i) = - (iv) ;
(i) = - (iv) ; 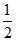 (iii) = - (ii)
(iii) = - (ii)
(i) = - (iii) ;
Create a free account to view solution
View Solution FreeMore Chemical Equilibrium Questions
Which of the following lines correctly show the temperature dependence of equilibrium constant, K, for an exothermic rea...For the equilibrium, A(g) B(g), ⇋H is −40 kJ/mol. If the ratio of the activation energies of the forward(Ef)...For the reaction H2(g) + I2(g) $\rightleftharpoons$2HI(g), the rate law expression is...One mole each of A and B and 3 moles each of C and D are placed in 1 L flask. If equilibrium constant is 2.25 for the re...1 mole each of H2(g) and I2(g) are introduced in a 1L evacuated vessel at 523K and equilibrium H2(g) + I2(g) ⇋ 2HI...