JEE Main | 2018Chemical EquilibriumHard
Question
Which of the following lines correctly show the temperature dependence of equilibrium constant, K, for an exothermic reaction ?
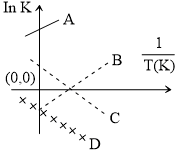
Options
A.
B and C
B.
C and D
C.
A and D
D.
A and B
Solution
From thermodynamics
lnk =
for exothermic reaction,
slope =
So from graph, line should be A & B.
Create a free account to view solution
View Solution FreeMore Chemical Equilibrium Questions
Suppose the gas phase isomerization reactions: A$\rightleftharpoons$B, A$\rightleftharpoons$C and B$\rightleftharpoons$C...Steam decomposes at high temperature according to the following equation.2H2O(g) $\rightleftharpoons$2H2(g) + O2(g); ΔHo...In the reaction N2(g) + 3H2(g) $\rightleftharpoons$2NH3(g) + Heat. One mole of N2 reacts with three moles of H2. If at e...For the reversible reaction, N2(g) + 3H2(g) ⇋ 2NH3(g) + Heat The equilibrium shifts in forward direction...In a flask, colourless N2O4 is in equilibrium with brown coloured NO2. At equilibrium, when the flask is heated at 100oC...