Chemical EquilibriumHard
Question
Select the reaction for which the equilibrium constant is written as [MX3]2 = Keq. [MX2]2 [X2]
Options
A.MX3 ⇋ MX2 +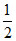 X2
X2
B.2MX3 ⇋ 2MX2 + X2
C.2MX2 + X2 ⇋ 2MX3
D.MX2 + 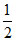 X2 ⇋ MX3
X2 ⇋ MX3
More Chemical Equilibrium Questions
A gaseous substance AB2(g) converts to AB(g) in the presence of solid A as AB2(g) + A(s)$\rightleftharpoons$ 2AB(g).The ...The position of equilibrium will shift in the given direction by the addition of inert gas at constant pressure in which...The equilibrium constant of the reaction SO2(g) + 1/2O2(g) ⇋ SO3(g) is 4 × 10-3 atm-1/2. The equilibrium cons...The process 2A(g) $\rightleftharpoons$A2(g) has KP = 8 × 108 atm−1. If ‘A’ atoms are taken at 1 atm pressure, then what ...An amount of 1 mole of PCl3(g) and 1 mole of PCl5(g) is taken in a vessel of 10 L capacity maintained at 400 K. At equil...