Chemical EquilibriumHard
Question
Ammonia dissociates into N2 and H2 such that degree of dissociation a is very less than 1 and equilibrium pressure is P0 then the value of a is [if Kp for 2NH3(g) ⇋ N2 (g) + 3H2(g) is 27 × 10-8 P02:
Options
A.10-4
B.4 × 10-4
C.0.02
D.can′t be calculated.
Solution
2NH3(g) ⇋ N2(g) + 3H2(g) Kp = 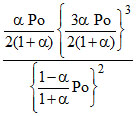
1 - α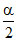
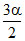
1- α ≃ 1 and 1 + α ≃ 1 ⇒ 27 × 10-8 P2o =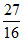 Po2 × α4
Po2 × α4
α = 2 × 10-2
1 - α
1- α ≃ 1 and 1 + α ≃ 1 ⇒ 27 × 10-8 P2o =
α = 2 × 10-2
Create a free account to view solution
View Solution FreeMore Chemical Equilibrium Questions
For a chemical reaction at the state of equilibrium, which of the following statement(s) is/are correct?...The condition suitable for forming atomic chlorine from molecular chlorine is...For the reaction SO2(g) + O2(g) ⇋ SO3(g), if KP = KC(RT)x where the symbols have usual meaning then the value of x...log + log RT = 0 is a relationship for the reaction :...Which of the following is not favourable for SO3 formation2SO2 (g) + O2 (g) ⇋ 2SO3 (g); ᐃH = - 45.0 kcal...