Chemical EquilibriumHard
Question
log 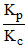 + log RT = 0 is a relationship for the reaction :
+ log RT = 0 is a relationship for the reaction :
Options
A.PCl5 ⇋ PCl3 + Cl2
B.2SO2 + O2 ⇋ 2SO3
C.H2 + I2 ⇋ 2HI
D.N2 + 3H2 ⇋ 2NH3
Solution
Using KP = KC (RT)ᐃng
so log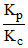 = ᐃng log RT
= ᐃng log RT
so, ᐃng = - 1.
so log
so, ᐃng = - 1.
Create a free account to view solution
View Solution FreeMore Chemical Equilibrium Questions
When CO2(g) is dissolved in water, then the following equilibrium is establishedCO2(aq) + 2H2O(l) $\rightleftharpoons$H3...PCl5 (molecular mass = M) dissociates into PCl3 and Cl2 as PCl5(g) $\rightleftharpoons$PCl3(g) + Cl2(g). If the total pr...ΔfGo for the formation of HI(g) from its gaseous elements is – 2.303 kcal/mol at 500 K. When the partial pressure of HI ...In an evacuated vessel of capacity 112 L, 4 moles of Ar(g) and 5 moles of PCl5(g) were introduced and the temperature is...For the reaction CaCO3(s) $\rightleftharpoons$CaO(s) + CO2(g), the value of KP is...