Chemical EquilibriumHard
Question
XY2 dissociates XY2(g) ⇋ XY(g) + Y(g). When the initial pressure of XY2 is 600 mm Hg, the total equilibrium pressure is 800 mm Hg. Calculate K for the reaction. Assuming that the volume of the system remains unchanged.
Options
A.50.0
B.100.0
C.166.6
D.400.0
Solution
XY2 ⇋ XY + Y
Initial P 0 0
Final P - x x x
Initial P = 600 mm Hg, Final P = 800 mm Hg
Total moles ∝ final P.
∴ P - x + x + x ∝ 800
600 - x + x + x ∝ 800
x = 200 mm Hg
K =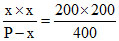 = 100 HG
= 100 HG
Initial P 0 0
Final P - x x x
Initial P = 600 mm Hg, Final P = 800 mm Hg
Total moles ∝ final P.
∴ P - x + x + x ∝ 800
600 - x + x + x ∝ 800
x = 200 mm Hg
K =
Create a free account to view solution
View Solution FreeMore Chemical Equilibrium Questions
Equilibrium constant for the reactions, 2 NO + O2 ⇋ 2 NO2 is KC1 ; NO2 + SO2 ⇋ SO3 + NO is KC2 and 2 SO3 ...The equilibrium constant for the reaction CO(g) + H2O(g) $\rightleftharpoons$CO2(g) + H2(g) is 3.0 at 500 K. In a 2.0 L ...For the reaction CaCO3(s) ⇋ CaO(s) + CO2(g), the pressure of CO2(g) depends on :...In a closed rigid vessel, the following equilibrium partial pressures are measured, where N2 = 100 mm, H2 = 400 mm and N...Two moles of an equimolar mixture of two alcohols R1–OH and R2–OH are esterified with one mole of acetic acid. If only 8...