Chemical EquilibriumHard
Question
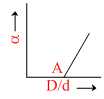
Before equilibrium is set-up for the chemical reaction N2O4(g) ⇋ 2NO2(g), vapour density d of the gaseous mixture was measured. If D is the theoretical value of vapour density, variation of a with D/d is given by the graph. What is value D/d at point A?

Options
A.0
B.0.5
C.1
D.1.5
More Chemical Equilibrium Questions
Iron fillings and water were placed in a 5 L vessel and sealed. The tank was heated to 1000oC. Upon analysis, the tank w...The value of KP for the equilibrium NH4HS(s) $\rightleftharpoons$NH3(g) + H2S(g) at a certain temperature and total 2 at...The equilibrium mixture for the reaction 2H2S(g)$\rightleftharpoons$ 2H2(g) + S2(g) has 1 mole of H2S, 0.20 mole of H2 a...Consider the following equilibrium in a closed container N2O4(g) ⇋ 2NO2(g) At a fixed temperature, the volume of t...For the chemical reaction 3X(g) + Y(g) $\rightleftharpoons$X3Y(g), the amount of X3Y at equilibrium is affected by...