Chemical EquilibriumHard
Question
In the above question, a varies with D/d according to :
Options
A.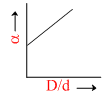

B.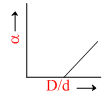

C.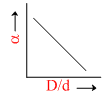

D.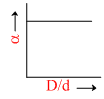

Solution
α = 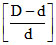 ; α =
; α = 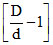 ;
; 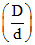 = α + 1.
= α + 1.
α =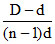 ; α =
; α = 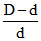 ; α =
; α = 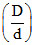 - 1.
- 1.
The point at which α = 0.
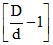 = 0 ; So
= 0 ; So 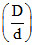 = 1.
= 1.
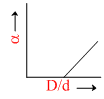
α =
The point at which α = 0.

Create a free account to view solution
View Solution FreeMore Chemical Equilibrium Questions
For the reversible reaction, N2(g) + 3H2(g) ⇋ 2NH3(g) + Heat The equilibrium shifts in forward direction...An industrial fuel, ′water gas′ , which consists of a mixture of H2 and CO can be made by passing steam over...For the reaction N2O4 (g) ⇋ 2NO2 (g), if percentage dissociation of N2O4 are 20%, 45%, 65% & 80%, then the sequenc...1 mole each of H2(g) and I2(g) are introduced in a 1L evacuated vessel at 523K and equilibrium H2(g) + I2(g) ⇋ 2HI...Carbon monoxide in water gas reacts with steam according to the following reaction.CO(g) + H2O(g) $\rightleftharpoons$CO...