Atomic StructureHard
Question
The electrons, identified by quantum numbers n and l, (i) n = 4, l = l (ii) n = 4, l = 0 (iii) n = 3, l = 2 and (iv) n = 3, l = 1 can be placed in order of increasing energy from the lowest to highest, as :
Options
A.(iv) < (ii) < ( iii) < (i)
B.(ii) < (iv) < ( i) < (iii)
C.(i) < (iii) < ( ii) < (iv)
D.(iii) < (i) < ( iv) < (ii)
Solution
Using (n + l)
Set : (i) (ii) (iii) (iv)
(n + l) : 5 4 5 4
Thus correct order of increasing energy is
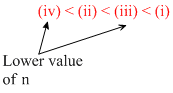
Set : (i) (ii) (iii) (iv)
(n + l) : 5 4 5 4
Thus correct order of increasing energy is
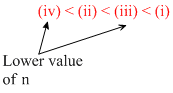
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
The ionization energy of He-atom in the ground state may be...The following charged particles accelerated from rest through the same potential difference are projected towards gold ...Calculate the wavelength (in nanometer) associated with a proton moving at 1.0 × 103ms-1 (Mass of proton = 1.67 ...The energy required by electrons, present in the first Bohr orbit of hydrogen atom to be excited to second Bohr orbit is...From a hydrogen discharge tube, only three photons are picked up. The energies of these three photons were 10.2, 12.1 an...