Atomic StructureHard
Question
The correct set od quantum numbers for the unpaired electron of chlorine atom is :
Options
A.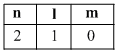

B.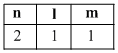

C.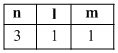

D.

Solution
17C = 1s2, 2s2, 2p6, 3s2, 3p5 ← unpaired electron lies in it
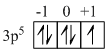
Thus n = 3, l = 1, m = 1
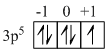
Thus n = 3, l = 1, m = 1
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
Which of the following is electronic configuration of Cu2+ (Z = 29) ?...A variable, opposite external potential (Eext) is applied to the cell Zn|Zn2+ (1 M) || Cu2+ (1M) | Cu, of potential 1.1 ...Photon having energy equivalent to the binding energy of 4th state of He+ atom is used to eject an electron from the met...The angular momentum of electron in ′d′ orbital is equal to :-...Choose the wrong one on the basis of Bohr′s theory :-...