Chemical EquilibriumHard
Question
In the reaction C(s) + CO2(g) ⇋ 2CO(g), the equilibrium pressure is 12 atm. If 50% of CO2 reacts then Kp will be :
Options
A.12 atm
B.16 atm
C.20 atm
D.24 atm
Solution
C(s) + CO2(g) ⇋ 2CO(g)
P - P/2 P =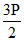 = 12
= 12
so KP =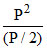 = 2P = 2 × 8 = 16 atm.
= 2P = 2 × 8 = 16 atm.
P - P/2 P =
so KP =
Create a free account to view solution
View Solution FreeMore Chemical Equilibrium Questions
For the reaction, 2NOBr(g) $\rightleftharpoons$2NO(g) + Br2(g), the ratio $\frac{K_{p}}{P}$, where P is the total pressu...In the preceeding problem, if [A+] and [AB2-] are y and x respectively, under equilibrium produced by adding the substan...At 444°C, HI is 30% dissociated. If initially 3 moles of HI are taken, then the number of moles of HI at equilibrium is...Rate of disappearance of the reactant ‘A’ in the reversible reaction A$\rightleftharpoons$B at two temperatures is given...Solid ammonium carbamate dissociates as: NH2COONH4(s) $\rightleftharpoons$2NH3(g) + CO3(g). In a closed vessel, solid am...