Chemical EquilibriumHard
Question
For the reaction H2(g) + I2(g) ⇋ 2HI(g)
Kc = 66.9 at 350oC and Kc = 50.0 at 448oC. The reaction has
Kc = 66.9 at 350oC and Kc = 50.0 at 448oC. The reaction has
Options
A.ᐃH = + ve
B.ᐃH = - ve
C.ᐃH = zero
D.ᐃH sign can not be determined
Solution
H2(g) + I2(g) ⇋ 2HI(g)
log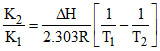
log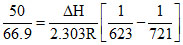
After calculation negative value of ᐃH is obtained.
log
log
After calculation negative value of ᐃH is obtained.
Create a free account to view solution
View Solution FreeMore Chemical Equilibrium Questions
Steam decomposes at high temperature according to the following equation.2H2O(g) $\rightleftharpoons$2H2(g) + O2(g); ΔHo...Consider the following equilibrium in a closed container. N2O4(g) $\rightleftharpoons$2NO2(g)At a fixed temperature, the...Which one of the following has lowest melting point :-...An aqueous solution of volume 500 ml, when the reaction 2Ag+ (aq) + Cu(s) $\rightleftharpoons$Cu2+ (aq) + 2Ag(s) reached...At chemical equilibrium, the...