Chemical EquilibriumHard
Question
The average person can see the red colour imparted by the complex [Fe(SCN)]2+ to an aqueous solution if the concentration of the complex is 6 × 10-6 M or greater. What minimum concentration of KSCN would be required to make it possible to detect 1 ppm (part per million) of Fe(III) in a natural water sample? The instability constant for Fe(SCN)2+ ⇋ Fe3+ + SCN- is 7.142 × 10-3.
Options
A.0.0036 M
B.0.0037 M
C.0.0035 M
D.None of these
Solution
Fe3+ + SCN- ⇋ [Fe(SCN)]2+ K = 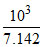
Initial conc.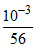 b 0
b 0
Final conc.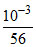 - 6 × 10-6 b - 6 × 10-6 6 × 10-6
- 6 × 10-6 b - 6 × 10-6 6 × 10-6
on solving b = 0.0036 M.
Initial conc.
Final conc.
on solving b = 0.0036 M.
Create a free account to view solution
View Solution FreeMore Chemical Equilibrium Questions
For the equilibrium SrCl2·6H2O(s) $\rightleftharpoons$SrCl2·2H2O(s) + 4H2O(g), KP = 8.1 × 10−7 atm4 at 27oC. If 1.642 L ...The reaction which proceeds towards completion in the forward direction is...At constant temperature, the equilibrium constant (Kp)for the decomposition reaction,N2O4 ⇋ 2NO2 is expressed by K...In an evacuated rigid vessel of volume V litre, one mole of solid ammonium carbonate (NH2CONH4) is taken and the vessel ...Phosphine decomposes at 300 K and 3 atmosphere as 4PH3(g) ⇋ P4(g) + 6H2(g)Calculate the vapour density of phosphin...