Chemical EquilibriumHard
Question
N2 and H2 are taken in 1 : 3 molar ratio in a closed vessel to attained the following equilibrium N2(g) + 3H2(g) ⇋ 2NH3(g) . Find Kp for reaction at total pressure of 2P if PN2 at equilibrium is P/3
Options
A.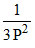
B.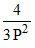
C.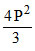
D.none
Solution
N2(g) + 3H2(g) ⇋ 2NH3(g)
At equilibrium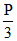 P
P 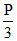 + P + PNH3 = 2P
+ P + PNH3 = 2P
PNH3 =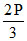 Kp =
Kp = 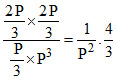 ⇒ Kp =
⇒ Kp = 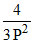
At equilibrium
PNH3 =
Create a free account to view solution
View Solution FreeMore Chemical Equilibrium Questions
Hydrolysis of sucrose gives :-Sucrose + H2O ⇋ Glucose + Fructose Equilibrium constant KC for the reaction is 2 ...Ammonia gas at 15 atm is introduced in a rigid vessel at 300 K. At equilibrium the total pressure of the vessel is found...In a vessel of 1.0 L capacity, O2(g) at 0.25 atm pressure and HCl(g) at 1.0 atm pressure are allowed to react in the pre...For the reaction 2NO2(g) ⇋ 2NO(g) + O2(g), (Kc = 1.8 × 10-6 at 184oC) (R = 0.0831 kJ / (mol.K) When KP and Kc...For a reaction,A → B ; if log10 K(sec-1) = 14 - the Arrheniums parameter and energy of activation for the reaction...