SolutionHard
Question
For chloroform and acetone or for a solution of chloroform and acetone if ps (observed (actual)) is compared with ps (Theoretical (Raoult)) then which of the following is /are true ?
Options
A.ps(actual) < ps(raoult)
B.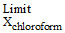 → 0 (poacetone - ps actual) = 0
→ 0 (poacetone - ps actual) = 0
C.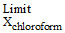 → (pochloroform - ps (actual)) = 0
→ (pochloroform - ps (actual)) = 0
D.poacetone > po chloroform near room temperature
Solution
CHCl3 + Acetone : Negative deviation from Raoult′s law.
So, pactual < pRaoult.
So, pactual < pRaoult.
Create a free account to view solution
View Solution FreeMore Solution Questions
How do we differentiate between Fe3+ and Cr3+ in group III?...The degree of dissociation (α) of a weak electrolyte, Ax By is related to van′t Hoff factor (i) by the expres...Some of the following gases are soluble in water due to formation of their ions : I : CO2 ; II : NH3 ; III : HCl ; IV : ...Dry air was passed successively through a solution of 5 g of a solute in 180 g of water and then through pure water. The...FeCl3 solution (side X) on reaction with K4[Fe(CN)6] (side Y) in aqueous solution gives blue colour. These are separated...