SolutionHard
Question
Dry air was passed successively through a solution of 5 g of a solute in 180 g of water and then through pure water. The loss in the weight of solution was 2.50 g and that of pure sovent 0.04 g. The molecular weight of the solute is
Options
A.31.25
B.3.125
C.312.5
D.None
Solution
Po - Ps ∝ loss in weight of water chamber and PS ∝ loss in weight of solution chamber.
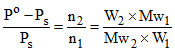
or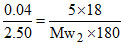
∴ Mw2 = 31.25
or
∴ Mw2 = 31.25
Create a free account to view solution
View Solution FreeMore Solution Questions
For which of the following pair, the heat of mixing, ΔHmix, is approximately zero?...For an ideal solution of A and B, YA is the mole fraction of A in the vapour phase at equilibrium. Which of the followin...Consider following cases :I : 2M CH3COOH solution in benzene at 27oC where there is dimer formation to the extent of 100...Which characterises the weak intermolecular forces of attraction in a liquid ?...At 35°C, the vapour pressure of CS2 is 512 mm Hg and that of acetone CH3COCH3 is 344 mm Hg. A solution of CS2 and aceton...