SolutionHard
Question
Consider following cases -
I : 2M CH3COOH solution in benzene at 27oC where there is dimer formation to the extent of 100%
II : 0.5 M KCl aq. solution at 27oC, which ionises 100%
Which is/are true statements(s) -
I : 2M CH3COOH solution in benzene at 27oC where there is dimer formation to the extent of 100%
II : 0.5 M KCl aq. solution at 27oC, which ionises 100%
Which is/are true statements(s) -
Options
A.both are isotonic
B.I is hypertonic
C.II is hypotonic
D.none is correct
Solution
πI = 2R × 300 × 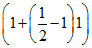 = 300 R
= 300 R
πII = 0.5 R × 300 × 2 = 300 R
πII = 0.5 R × 300 × 2 = 300 R
Create a free account to view solution
View Solution FreeMore Solution Questions
Which of the following compounds can be used as antifreeze in automobile radiators?...The solubility of solid silver chromate, Ag2CrO4 is determined in three solvents. (Ksp = 9 × 10-12)(i) Pure water (...In which case, van′t Hoff factor i remains unchanged ? [Assume common complexes of these ions]...Which of the following combinations are correct for a binary solution, in which the solute as well as solvent are liquid...Freezing point lowering expression is ᐃTf = Kfm (molality)Which of the following assumptions are considered for th...