SolutionHard
Question
Given at 350 K pAo = 300 torr and pBo = 800 torr, the composition of the mixture having a normal boiling point of 350 K is :
Options
A.XA = 0.08
B.XA = 0.06
C.XA = 0.04
D.XA = 0.02
Solution
760 = 300 XA + 800 (1 - XA)
⇒ 760 = 800 - 500 XA
⇒ 500 XA = 40
∴ XA =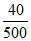 = 0.08.
= 0.08.
⇒ 760 = 800 - 500 XA
⇒ 500 XA = 40
∴ XA =
Create a free account to view solution
View Solution FreeMore Solution Questions
Consider following solutions :I : 1 M aqueous glucose solution II : 1M aqueous sodium chloride solution III : 1M aqueous...0.01 M solution of KCl and BaCl2 are prepared in water. The freezing point of KCl is found to be 2oC....What is molarity of HCl in a solution prepared by dissolving 5.5g HCl in 200 g ethanol. If the density of the solution i...A solution of Na2S2O3 is standardized iodometrically against 0.167 g of KBrO3 where BrO3- changesto Br-. This process re...Select incorrect statement :...