SolutionHard
Question
25 mL of an aqueous solution of KCl was found to requires 20 mL of 1M AgNO3 solution when titrated using a K2CrO4 as indicator. Depression in freezing point of KCl solution with 100% ionization will be
[Kf = 2.0o mol-1 kg and molarity = molality]
[Kf = 2.0o mol-1 kg and molarity = molality]
Options
A.20/45
B.80/45
C.40/45
D.160/45
Solution
KCl + AgNO3 → AgCl + KNO3
0.8 × 25 1 × 20 0 0
(mili mole) (mili mole)
0 0 20 20
Mole of KNO3 in solutions =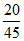
ᐃTf = i × m × Kf
ᐃTf = 2 ×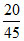 × 2 =
× 2 = 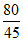
0.8 × 25 1 × 20 0 0
(mili mole) (mili mole)
0 0 20 20
Mole of KNO3 in solutions =
ᐃTf = i × m × Kf
ᐃTf = 2 ×
Create a free account to view solution
View Solution FreeMore Solution Questions
Insulin (C2H10O5)n is dissolved in a suitable solvent and the osmotic pressure p of the solution of various concentratio...A 5% solution (by mass) of cane sugar in water has freezing point of 271 K and freezing point of pure water is 273.15 K....Thevan′t Hoff factor, i for a compound which undergoes dissociation in one solvent and association in other solven...The osmotic pressures of 0.010 M solutions of KI and of sucrose (C12H22O11) are 0.432 atm and 0.24 atm respectively. The...A teacher one day pointed out to his students the preculia fact that water is unique liquid which freezes exactly at 0o ...