SolutionHard
Question
A 5% solution (by mass) of cane sugar in water has freezing point of 271 K and freezing point of pure water is 273.15 K. The freezing point of 5% solution (by mass) of glucose in water is
Options
A.271 K
B.273.15 K
C.269.07
D.277.23 K
Solution
From the relation
ᐃTf = Kf ×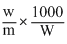
It is obvious that
ᐃTf ∝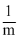
∴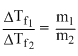
Cane sugar solution Glucose solution
ᐃTf1 = 273.15 = 271 ᐃTf2
m1 = 324 m2 = 180
Hence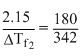
ᐃTf2 = 4.085 K
So freezing point of glucose solution
= 273.15 - 4.085
= 269.05 K
ᐃTf = Kf ×
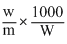
It is obvious that
ᐃTf ∝
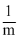
∴
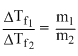
Cane sugar solution Glucose solution
ᐃTf1 = 273.15 = 271 ᐃTf2
m1 = 324 m2 = 180
Hence
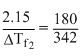
ᐃTf2 = 4.085 K
So freezing point of glucose solution
= 273.15 - 4.085
= 269.05 K
Create a free account to view solution
View Solution FreeMore Solution Questions
A 0.001 molal solution of a complex MA8 in water has the freezing point of −0.0054°C. Assuming 100% ionization of the co...One molal solution of benzoic acid in benzene boils at 81.53°C. The normal boiling point of benzene is 80.10°C. Assuming...For an ideal solution containing two liquid components A and B, the Gibb’s free energy of mixing is minimum, when the mo...Aqueous solution of acedic acid contains :-...The relationship between osmotic pressure at 273 K when 10 g glucose (P1) ,10 g urea (P2) and 10 g sucrose (P3) are diss...