SolutionHard
Question
PtCl4. 6H2O can exist as a hydrated complex 1 molal aq. solution has depression in freezing point of 3.72o. Assume 100% ionisation and Kf(H2O) = 1.86o mol-1 kg, then complex is -
Options
A.[Pt(H2O)6]Cl4
B.[Pt(H2O)4Cl2]Cl2 . 2H2O
C.[Pt(H2O)3Cl3]Cl . 3H2O
D.[Pt(H2O)2Cl4]. 4H2O
Solution
ᐃTf = i × m × kf
i =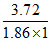 = 2
= 2
So salt is (Pt (H2O)3 Cl3) Cl. 3H2O.
i =
So salt is (Pt (H2O)3 Cl3) Cl. 3H2O.
Create a free account to view solution
View Solution FreeMore Solution Questions
An ether is more volatile than an alcohol having the same molecular formula. This is due to...The vapour pressure of benzene at 30oC is 121.8 mm. By adding 15g of non-volatile solute in 250 g of benzene, its molecu...Select incorrect statement :...Fog is a colloidal solution of:...A solution is prepared by dissolvint 24.5 g of sodium hydroxide in distilled water to give 1L solution. The molarityof N...