SolutionHard
Question
A graph plotted between 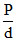 vs , d (where P is osmotic pressure of solution of a solute of mol. wt. m and d is its density at temperature T. Pick out the correct statements about the plots :
vs , d (where P is osmotic pressure of solution of a solute of mol. wt. m and d is its density at temperature T. Pick out the correct statements about the plots :
Options
A.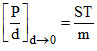
B.The intercept of the plot 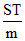
C.The intercept of the plot = 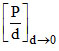
D.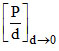 is independent of temperature
is independent of temperature
More Solution Questions
H2S on passing through ammonical solution gives white ppt. The white ppt. is of :-...Among the following the surfactant that will form micelles in aqueous solution at the lowest molar concentration at ambi...Thevan′t Hoff factor, i for a compound which undergoes dissociation in one solvent and association in other solven...An aqueous solution of a salt X turns blood red on treatment with SCN− and blue on treatment with K4[Fe(CN)6]. X a...Water and chlorobenzene are immiscible liquids. Their mixture boils at 90oC under a reduced pressure of 7.82 × 104 ...