SolutionHard
Question
Ethylene glycol is used as an antifreeze in a cold climate. Mass of ethylene glycol which should be added to 4 kg of water to prevent it from freezing at - 6oC will be :
[f K for water = 1.86 K kg mol ″1 , and molar mass of ethylene glycol = 62g/ mol ]
[f K for water = 1.86 K kg mol ″1 , and molar mass of ethylene glycol = 62g/ mol ]
Options
A.204.30g
B.400.00 g
C.304.60 g
D.804.32g
Solution
ᐃTf = Kf × m = Kf × 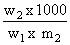
w1 & w2 = wt of solvent & solute respecting
m2 = mw of solute
ᐃTf = 0o - ( - 6o) = 6 1.86 ×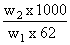
Therefore w2 = 800g
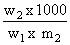
w1 & w2 = wt of solvent & solute respecting
m2 = mw of solute
ᐃTf = 0o - ( - 6o) = 6 1.86 ×
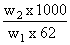
Therefore w2 = 800g
Create a free account to view solution
View Solution FreeMore Solution Questions
Equal volumes of M/20 glucose solution at 300 K and M/20 sucrose solution at 300 K are mixed without change in temperatu...At a constant temperature, DS will be maximum for which of the following processes :...During depression of freezing point in a solution the following are in equilibrium :...Relative decrease in vapour pressure of an aqueous solution containing 2 moles [Cu(NH3)3Cl] Cl in 3 moles H2O is 0.50. O...For a dilute solution containing 2.5 g of a non-volatile non-electrolyte solute in 100 g of water, the elevation in boil...