Gaseous StateHard
Question
A bottle of dry ammonia and a bottle of dry hydrogen chloride connected through a long tube are opened simultaneously at both ends the white ammonium chloride ring first formed will be :
Options
A.at the centre of the tube
B.near the hydrogen chloride bottle
C.near the ammonia bottle
D.throughout the length of the tube
Solution
Rate of diffusion 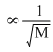
∴ MHCL > MNH3
∴ 4HCL diffuses at slower rate which NH3 diffuses at higher rate. So NH4Cl is formed near the HCL bottle.
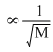
∴ MHCL > MNH3
∴ 4HCL diffuses at slower rate which NH3 diffuses at higher rate. So NH4Cl is formed near the HCL bottle.
Create a free account to view solution
View Solution FreeMore Gaseous State Questions
Which the temperature is increased, surface tension of water :...A gas X diffuses three times faster than another gas Y. The ratio of their densities is :-...What volume will be occupied by a sample of gas at 40oC and 720 mm μg if its volume at 20oC and 750 mm μg was ...NH3 gas is liquefied more easily than N2. Hence:...The rate of diffusion of SO2, CO2, PCl3 and SO3 are in the following order...