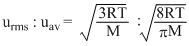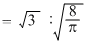Gaseous StateHard
Question
The ratio of root mean square velocity to average velocity of a gas molecule at a particular temperature is :
Options
A.1.086 : 1
B.1 : 1.086
C.2 : 1.086
D.1.086 : 2
More Gaseous State Questions
The vander waal gas constant ′a′ is given by...Two flasks of equal volume are connected by a narrow tube (of negligible volume) all at 27oC and contain 0.70 mole of H2...1 mol of a gaseous aliphahatic compound CnH3nOm is completely burnt in an excess of oxygen. The contraction in volume is...A gas is allowed to expand in a well insulated container against a constant external pressure of 2.5atm from an initial ...The volume of CO2 produced by the combustion of 40 ml of gaseous acetone in excess of oxygen is...