Redox and Equivalent ConceptHard
Question
The normality of 0.3 M phosphorous acid (H3PO3) is :
Options
A.0.1
B.0.9
C.0.3
D.0.6
Solution
0.6 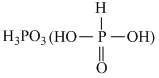 is dibasic acid.
is dibasic acid.
∴ N = M × is dibasic acid.
= 0.3 × 2 = 0.6
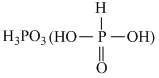 is dibasic acid.
is dibasic acid.∴ N = M × is dibasic acid.
= 0.3 × 2 = 0.6
Create a free account to view solution
View Solution FreeMore Redox and Equivalent Concept Questions
Acetic acid on chlorination yields trichloroacetic acid. Its equivalent weight will be...Reduction of the metal centre in aqueous permanganate ion involves...When a metal carbonate is treated with excess of dilute sulphuric acid, the weight of metal sulphate formed is 1.43 time...The co-ordination number and oxidation state of Cr in K3[Cr(C2O4)3] are respectively :-...A redox reaction is...