Mole ConceptHard
Question
Depression of freezing point of 0.01 molal aq. CH3COOH solution is 0.02046. 1 molal urea solution freezes at -1.86oC. Assuming molality equal to molarity, pH of CH3COOH solution is
Options
A.2
B.3
C.3
D.4.2
Solution
For urea ᐃTf = i Kfm
1.86 = 1 × Kf × 1
Kf = 1.86
Now for acetic acid ⇒ ᐃTf = i Kf m
0.02046 = i × 1.86 × 0.01
i = 1.1
Now α =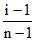 ⇒ α =
⇒ α = 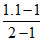
α = 0.1
H+ = C α = 0.01 × 0.1 = 10-3
pH = 3
1.86 = 1 × Kf × 1
Kf = 1.86
Now for acetic acid ⇒ ᐃTf = i Kf m
0.02046 = i × 1.86 × 0.01
i = 1.1
Now α =
α = 0.1
H+ = C α = 0.01 × 0.1 = 10-3
pH = 3
Create a free account to view solution
View Solution FreeMore Mole Concept Questions
Which pair of molecule have identical shape :-...Which of the following criteria does not pertain to facillitated transport ?...Density of ozone relative to oxygen is under the same temperature and pressure...2KI + I2 + 22 HNO3 → 2HIO3 + 2KIO3 + 22NO2 + 10H2OIf 3 mole of KI & 2 moles I2 are reacted with excess of HNO3. Vo...For the complete combustion of 0.2 moles of the sugar, how many moles of oxygen is required ?...