Coordination CompoundHard
Question
Which of the following is/are paramagnetic ?
Options
A.[Fe(CN)6]4-
B.[Ni(CO)4]
C.[Ni(CN)4]2-
D.[CoF6]3-
Solution
(A) [Fe(CN)6]4- d6 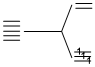 CN- is strong field lignad.
CN- is strong field lignad.
(B) [Ni(CO)4] d10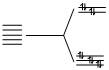 CO is strong field lignad.
CO is strong field lignad.
(C) [Ni(CN)4]2- ; Ni2+ has 3d8 configuration and CN- is strong field lignad. So, complex is square planar and diamagnetic.
(D) [CoF6]3- d6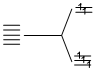 F- being weak field ligand does not allow pairing.
F- being weak field ligand does not allow pairing.
 CN- is strong field lignad.
CN- is strong field lignad.(B) [Ni(CO)4] d10
 CO is strong field lignad.
CO is strong field lignad.(C) [Ni(CN)4]2- ; Ni2+ has 3d8 configuration and CN- is strong field lignad. So, complex is square planar and diamagnetic.
(D) [CoF6]3- d6
 F- being weak field ligand does not allow pairing.
F- being weak field ligand does not allow pairing. Create a free account to view solution
View Solution FreeMore Coordination Compound Questions
Which of the following statements is correct?...Which of the following pairs will show the same magnetic moment (′spin only′)?...Low spin complex is formed by :...Which of the following complexes are low spin and diamagnetic ?(1) K4[Os(CN)6] (2) [Mo(CO)6] (3) [Mn(CN)6]4-Select the c...The IUPAC name for the complex [Co(NO2)(NH3)5]Cl2 is...