Nitrogen Containing CompoundHard
Question
The correct order of the basicities of the following compounds is :
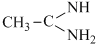 , CH3 - CH2 - NH2, (CH3)2NH,
, CH3 - CH2 - NH2, (CH3)2NH, 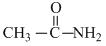
1 2 3 4
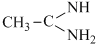 , CH3 - CH2 - NH2, (CH3)2NH,
, CH3 - CH2 - NH2, (CH3)2NH, 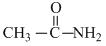
1 2 3 4
Options
A.2 > 1 > 3 > 4
B.1 > 3 > 2 > 4
C.3 > 1 > 2 > 4
D.1 > 2 > 3 > 4
Solution
The correct order is :
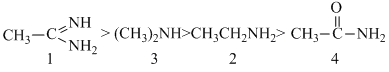
(1) is most basic because the cation obtained by its protonation (i.e, its conjugate acid) is highly stabilized by resonance.

(4) is least baisc because lone pair of -NH2 gp. intercts with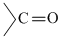 gp. and thus less available for protonation.
gp. and thus less available for protonation.
(3) and (4) both are aliphatic anines but (3), a secondary amine is more basic than (2), a primary amine is more basic than (2), a primary amine, due to more + I effect in (3)
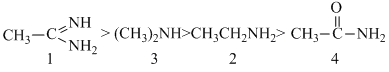
(1) is most basic because the cation obtained by its protonation (i.e, its conjugate acid) is highly stabilized by resonance.

(4) is least baisc because lone pair of -NH2 gp. intercts with
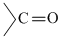 gp. and thus less available for protonation.
gp. and thus less available for protonation.(3) and (4) both are aliphatic anines but (3), a secondary amine is more basic than (2), a primary amine is more basic than (2), a primary amine, due to more + I effect in (3)
Create a free account to view solution
View Solution FreeMore Nitrogen Containing Compound Questions
Which of the following compounds possesses a chiral nitrogen atom ?...Which of the following compounds on hydrolysis yields a carboxylic acid and a secondary amine ?...The compound which give an oily nitrosoamine on reaction with acid at low temperature, is...How many products will be obtained when propane is subjected to vapour phase nitration ?...A compound with molecular mass 180 is acylated with CH3COCl to get a compound with molecular mass 390. The number of ami...