Nitrogen Containing CompoundHard
Question
A compound with molecular mass 180 is acylated with CH3COCl to get a compound with molecular mass 390. The number of amino groups present per molecule of the former compound is :-
Options
A.2
B.5
C.4
D.6
Solution
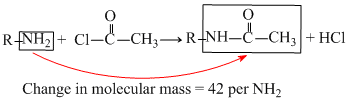
180 molecular mass after acylation gives compound of molecular mass 390.
Change in molecular mass = 390 - 180 = 210, so number of NH2 group
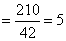
Create a free account to view solution
View Solution Free