Coordination CompoundHard
Question
Which of the following statements is false ?
Options
A.Complex of Pt (+II) and Au (+III) are square planar - including those with weak field ligands such as halide ions.
B.In tetrahedral complex , the t2g orbitals are nearer to the direction of the ligands.
C.For do, d5 and d10 arrangements the CFSE is zero in both octahedral and tetrahedral complexes with weak field ligands.
D.None.
Solution
(A) Pt (+II) and Au (+III) with 5d8 configuration have greater CFSE and thus favours square planar geometry (i.e. dsp2 hybridisation) with weak as well as strong field ligands.
(B) In tetrahedral complex , the t2g orbitals are nearer to the direction of the ligands. Hence they have higher energies than that of eg orbitals (for detail refer text).
(C) 3d5,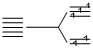 ; CFSE (weak filed ligands) = - 0.6 × 2 + 0.4 × 3 = 0 ᐃt
; CFSE (weak filed ligands) = - 0.6 × 2 + 0.4 × 3 = 0 ᐃt
3d5,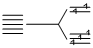 ; CFSE (weak filed ligands) = - 0.4 × 3 + 0.6 × 2 = 0 ᐃ0.
; CFSE (weak filed ligands) = - 0.4 × 3 + 0.6 × 2 = 0 ᐃ0.
Similarly for d0 and d10 configurations the CFSE is zero in both octahedral and tetrahedral complexes with weak field ligands.
(B) In tetrahedral complex , the t2g orbitals are nearer to the direction of the ligands. Hence they have higher energies than that of eg orbitals (for detail refer text).
(C) 3d5,
 ; CFSE (weak filed ligands) = - 0.6 × 2 + 0.4 × 3 = 0 ᐃt
; CFSE (weak filed ligands) = - 0.6 × 2 + 0.4 × 3 = 0 ᐃt 3d5,
 ; CFSE (weak filed ligands) = - 0.4 × 3 + 0.6 × 2 = 0 ᐃ0.
; CFSE (weak filed ligands) = - 0.4 × 3 + 0.6 × 2 = 0 ᐃ0.Similarly for d0 and d10 configurations the CFSE is zero in both octahedral and tetrahedral complexes with weak field ligands.
Create a free account to view solution
View Solution FreeMore Coordination Compound Questions
Trioxalatoaluminate(III) and tetrafluoro-borate(III) ions are:...Which of the following pairs of compounds are positional isomers ?...Which of the following statements is not correct?...The only cations present in a slightly acidic solution are Fe3+, Zn2+ and Cu2+. the reagent that when added in excess to...What will be the correct order for the wave length of light observed for the following ?(I) [Ni(NO2)6]4- , (II) [ Ni (NH...