Coordination CompoundHard
Question
A coordination compound of cobalt has the molecular formula containing five ammonia molecules, one nitro group and two chlorine atoms for one cobalt atom. One mole of this compound produces three moles of ions in an aqueous solution. The aqueous solution on treatment with an excess of AgNO3 gives two moles of AgCl as a precipitate. The formula of this complex would be
Options
A.[Co(NH3)4(NO2)Cl] [(NH3)Cl]
B.[Co(NH3)5Cl] [Cl NO2]
C.[Co(NH3)5(NO2)]Cl2
D.[Co (NH3)5] [(NO2)2Cl2]
Solution
[Co(NH3)5(NO2)]Cl2 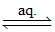 [Co(NH3)5(NO2)]2+ (aq) + 2Cl-(aq) (no. of ions = 3)
[Co(NH3)5(NO2)]2+ (aq) + 2Cl-(aq) (no. of ions = 3)
2Ag+ + 2Cl- → 2AgCl ↓ (white).
2Ag+ + 2Cl- → 2AgCl ↓ (white).
Create a free account to view solution
View Solution FreeMore Coordination Compound Questions
Concentrated H2SO4 will not dehydrate the following complex :...Which of the following facts about the complex [Cr(NH3)6]Cl3 is wrong ?...What will be the theoretical value of ′spin only′ magnetic moment when Fe(SCN)3 reacts with a solution conta...Which of the following is greatest paramagnetic ?...Which ligand forms chelates :-...