Coordination CompoundHard
Question
Which of the following statements is/are incorrect for the complex [Cr(H2O)6]Cl3?
Options
A.It has a magnetic moment of 3.83 BM.
B.The distribution of 3d electrons is 3dxy1, 3dyz1, 3dzx1
C.The ligand has satisfied both primary and secondary valencies of chromium.
D.It shows ionization as well as hydrate isomerism.
Solution
(A) The 3d3 electron configuration always have three unpaired electrons with weak field as well as strong field ligands.
(B) 3d3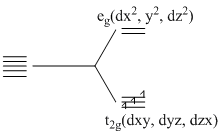
(C) H2O being neutral molecule satisfies only secondary valencies.
(D) It shows hydrate isomerism and not ionisation isomerism because anionic ligand is not present in coordination sphere.
(B) 3d3
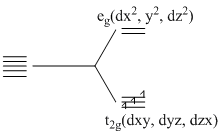
(C) H2O being neutral molecule satisfies only secondary valencies.
(D) It shows hydrate isomerism and not ionisation isomerism because anionic ligand is not present in coordination sphere.
Create a free account to view solution
View Solution FreeMore Coordination Compound Questions
Of the following complex ions, the one that probably has the largest overall formation constant, Kf, is :...Which of the following isomers of [M(NH3)2Cl2] would react with silver oxalate (Ag2C2O4) ?...Which of the following statements is not true?...In which of the following coordination entities, the magnitude of ∆0 [CFSE in octahedral field] will be maximum?:...Which of the following is/are inner orbital complex(es) as well as dimagentic in nature ?...