Coordination CompoundHard
Question
The complex [Pt(NH3)4]2+ has ..... structure :
Options
A.square planar
B.tetrahedral
C.pyramidal
D.pentagonal
Solution
5d8 configuration have higher CFSE and the complex is thus square planar and diamagnetic.
Pt2+, [Xe]4f14 5d8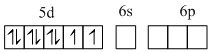
[Pt(NH3)4]2-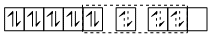
dsp2 hybrid orbitals
Pt2+, [Xe]4f14 5d8

[Pt(NH3)4]2-
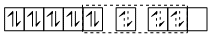
dsp2 hybrid orbitals
Create a free account to view solution
View Solution FreeMore Coordination Compound Questions
Among the following ions, which one has the highest paramagnetism ?...During estimation of nickel, we prepare nickel dimethylglyoxime, a scarlet solid. Thiscompound is...In which of the following pairs both the copmplexes show optical isomerism?...Which of the following is a bidentate, unsymmaterical, anionic ligand :-...In which of the following cases, the stability of two oxidation states is correctly represented...