Coordination CompoundHard
Question
Among the following ions, which one has the highest paramagnetism ?
Options
A.[Cr(H2O)6]3+
B.[Fe(H2O)6]2+
C.[Cu(H2O)6]2+
D.[Zn(H2O)6]2+
Solution
(A) Cr3+, 3d3 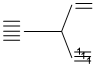
n = 3.
(B) Fe2+, 3d5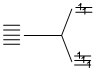
n = 5.
It has maximum number of unpaired electrons.
(C) Cu2+, 3d9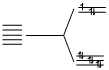
n = 0.
(D) Zn2+, 3d10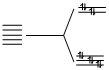
n = 0.

n = 3.
(B) Fe2+, 3d5

n = 5.
It has maximum number of unpaired electrons.
(C) Cu2+, 3d9
n = 0.
(D) Zn2+, 3d10

n = 0.
Create a free account to view solution
View Solution FreeMore Coordination Compound Questions
Coordination compounds have great importance in biological systems. In this context which of the following statements is...Which of the following is/are paramagnetic ?...Which of the following is correctly matched?...What is incorrect about homoleptic metal carbonyls?...The compounds containing complex oxyanion CrO42- are intensely yellow coloured be cause...