Coordination CompoundHard
Question
A compound is made by mixing cobalt (III) nitrite and potassium nitrite solutions in the ratio of 1 : 3. The aqueous solution of the compound showed 4 particles per molecule whereas molar conductivity reveals the presence of six electrical charges. The formula of the compound is :
Options
A.Co(NO2)3 . 2KNO2
B.Co(NO2)3 . 3KNO2
C.K3[Co(NO2)6]
D.K[Co(NO2)4]
Solution
Co(NO2)3 + 3KNO2 → K3[Co(NO2)6] 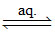 3K+ + [Co(NO2)6]3- (total six electric charge)
3K+ + [Co(NO2)6]3- (total six electric charge)
3 : 1 electrolyte.
3 : 1 electrolyte.
Create a free account to view solution
View Solution FreeMore Coordination Compound Questions
A solution containing 2.675 g of CoCl3.6 NH3 (molar mass = 267.5 g mol-1) is passed through a cation exchanger. The chlo...Among the following, the compound that is both paramagnetic and coloured is...Which of the following has an optical isomer ?...Among TiF62–, CoF63– , Cu2Cl2 and – NiCl42– the colourless species are:...Of the following complex ions, which is diamagnetic in nature ?...